- CEA
- CNRS |
- CEA Paris-Saclay |
- DRF Internet
- Iramis |
- LLB
- LLB intranet |
- VPN (Σigma - THOT - HORUS) |
- Webmail : Intra Extra
-
|

Laboratoire Léon Brillouin
UMR12 CEA-CNRS, Bât. 563 CEA Saclay
91191 Gif sur Yvette Cedex, France
+33-169085241 llb-sec@cea.fr
 Properties of ZrNi5 deuteride synthesized under high pressure studied by neutron diffraction and first principles calculations
Properties of ZrNi5 deuteride synthesized under high pressure studied by neutron diffraction and first principles calculations

V. Paul-Boncour, S.M. Filipek, J.-C. Crivello, F. Couturas, A. Morawski
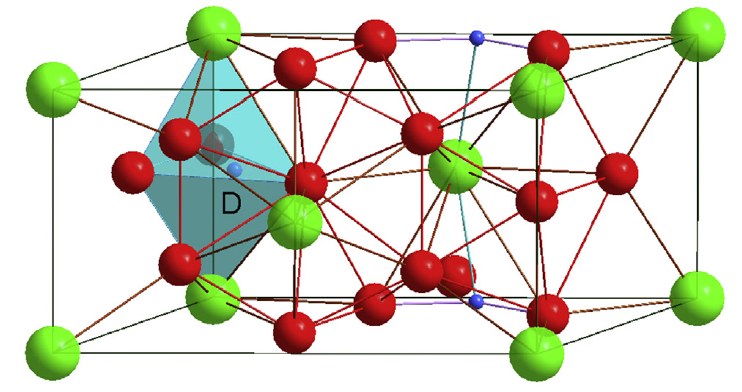
ZrNi5 deuteride was synthesized at a high deuterium pressure of 0.9 GPa and at 373 K. Neutron diffraction study has revealed that ZrNi5D0.96(4) crystallizes in a monoclinic structure (Cm space group) between 1.8 and 220 K. The D atoms are located in one trigonal bipyramid Zr2Ni3 interstitial site. Between 220 and 265 K a first order transition from the ordered monoclinic structure towards a disordered cubic structure is observed. In addition, a small deuterium desorption was observed after several hours at 240 K. Above 270 K, the compound is fully cubic and an accelerated deuterium desorption is observed within a two phase transformation. The kinetic of monoclinic-cubic transformation followed at 240 K is very slow, and the structural transformation is found reversible upon cooling. First principles calculations were performed in the frame of the Density Functional Theory (DFT) to understand the formation of the low temperature ZrNi5H1 hydride and its vibration properties.
