Coll. G. Sarret*, J. Covès°,
* Environmental Geochemistry Group, LGIT, University of Grenoble and CNRS, BP 53, 38041 Grenoble, Cedex 9, France.
° Institut de Biologie Structurale - J.-P. Ebel, Laboratoire des Protéines Membranaires, 41 rue Jules Horowitz, 38027 Grenoble Cedex, France.
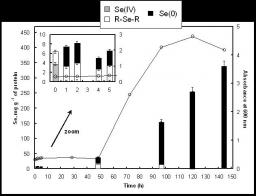
Ralstonia metallidurans CH34, a soil bacterium resistant to a variety of metals, is known to reduce selenite to intracellular granules of elemental selenium (Se0). We have studied the kinetics of selenite (SeIV) and selenate (SeVI) accumulation and used X-ray absorption spectroscopy to identify the accumulated form of selenate, as well as possible chemical intermediates during the transformation of these two oxyanions. When introduced during the lag phase, the presence of selenite increased the duration of this phase, as previously observed. Selenite introduction was followed by a period of slow uptake, during which the bacteria contained Se0 and alkyl selenide in equivalent proportions. This suggests that two reactions with similar kinetics take place: an assimilatory pathway leading to alkyl selenide, and a slow detoxification pathway leading to Se0. Subsequently, selenite uptake strongly increased (up to 340 mg Se per g of proteins), and Se0 was the predominant transformation product, suggesting an activation of selenite transport and reduction systems after several hours of contact. Exposure to selenate did not induce an increase in the lag phase duration and the bacteria accumulated approximately 25 fold less Se than when exposed to selenite. SeIV was detected as transient species in the first 12 hours after selenate introduction, Se0 also occurred as minor species, and the major accumulated form was alkyl selenide. Thus, in the present experimental conditions selenate mostly follows an assimilatory pathway, and the reduction pathway is not activated upon selenate exposure. These results show that R. metallidurans CH34 may be suitable for the remediation of selenite - but not selenate - contaminated environments.
This study illustrates the potential of XANES spectroscopy combined with elemental analyses, which enable the quantification of Se species. This spectroscopic approach is complementary to analytical speciation techniques such as liquid or ionic chromatography, or electrospray mass spectrometry, which are better suited to identify individual molecules.


 Physics and chemistry for life sciences and the environment › Human and Environmental Toxicology
Physics and chemistry for life sciences and the environment › Human and Environmental Toxicology 