Laboratoire Léon Brillouin
UMR12 CEA-CNRS, Bât. 563 CEA Saclay
91191 Gif sur Yvette Cedex, France
+33-169085241 llb-sec@cea.fr
Laboratoire Léon Brillouin
UMR12 CEA-CNRS, Bât. 563 CEA Saclay
91191 Gif sur Yvette Cedex, France
+33-169085241 llb-sec@cea.fr


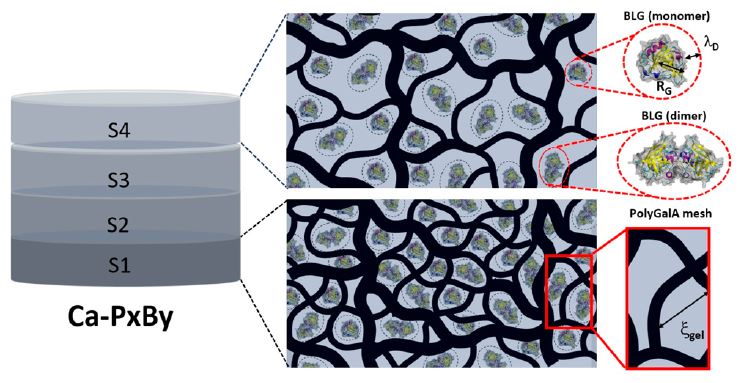
Schematic representation of the mesoscopic and local
structures of the mixed hydrogels. BLG proteins entrapped in the gel keep their native structure and exist in both monomeric and dimeric forms.
Aline Maire du Poset, Mikaela Börjesson, Céline Rameau, Claire Madeleine-Perdrillat, Adrien Lerbret, Camille Loupiac, Fabrice Cousin, Ali Assifaoui
We show here how the structure of polygalacturonate (polyGalA) hydrogels cross-linked by Ca2+ cations via external gelation controls the loading and release rate of beta-lactoglobulin (BLG), a globular protein. Hydrogels prepared from a polyGalA/BLG solution are found to be similar to those obtained from a polyGalA solution in our previous study (Maire du Poset et al. Biomacromolecules 2019, 20 (7), 2864–2872): they exhibit similar transparencies and gradients of mechanical properties and polyGalA concentrations. The nominal BLG/polyGalA ratio of the mixtures is almost recovered within the whole mixed hydrogel despite such strong concentration gradients, except in the part of the hydrogels with the largest mesh size, where more BLG proteins are present. This gradient enables one to tune the amount of protein loaded within the hydrogel. At a local scale, the proteins are distributed evenly within the hydrogel network, as shown by small-angle neutron scattering (SANS). The release of proteins from hydrogels is driven by Fickian diffusion, and the release rate increases with the mesh size of the network, with a characteristic time of a few hours. The specific structure of these polysaccharide-based hydrogels allows for control of both the dosage and the release rate of the loaded protein and makes them good candidates for use as oral controlled-delivery systems.
https://dx.doi.org/10.1021/acs.biomac.9b01722
• › Nanostructures et biomolécules : biomédecine et nanotoxicité / Nanostructures and biomolecules: biomedicine and nanotoxicity  Physique et chimie pour le vivant et l’environnement › Physique et vivant / Physics and life
Physique et chimie pour le vivant et l’environnement › Physique et vivant / Physics and life  Physique et chimie pour le vivant et l’environnement › Biophysique au LLB
Physique et chimie pour le vivant et l’environnement › Biophysique au LLB  Physics and chemistry for life sciences and the environment
Physics and chemistry for life sciences and the environment
• Laboratoire Léon Brillouin (LLB) • Leon Brillouin Laboratory (LLB)
• Groupe "Matière molle et biophysique" - MMB • Soft Complex Matter
